I’ve mentioned hydrocolloids at several occasions earlier in the blog, and today I found an interesting recipe I would like to share. Put simple, hydrocolloids are compounds that form gels when mixed with water. One particular hydrocolloid is methyl cellulose whose chemical structure is as follows:
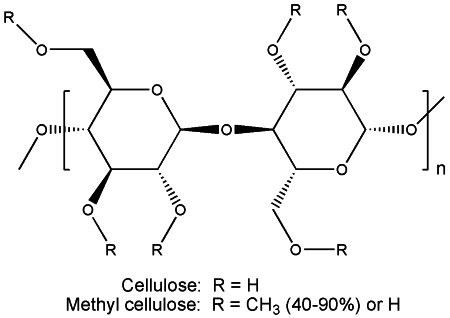
Methyl cellulose is made from cellulose. Methyl celluloses are available with varying degrees of methyl substitution. Typically 40-90% of the hydroxy groups are methylated. Often the degree of substitution (DS) is given as the average number of hydroxy groups that have been methylated per anhydroglucose unit, so the maximum DS is 3. The solubility in water decreases with increasing methyl substitution. One interesting property of methyl cellulose is the fact that it dissolves readily in cold water, but solidifies when you heat it (such gels are often referred to as thermoreversible). Using this property it is possible to make a hot “ice cream” that melts as it cools down. Does this sound weird? Here’s a recipe from Ideas in food so you can try it at home:
Hot Vanilla Ice Cream
306 g whole milk yogurt
230 g cream cheese
80 g agave nectar
154 g water
1 Bourbon vanilla bean scraped
pinch of sea salt
11.55g Methocel food gum (SGA150)In a blender puree together the yogurt, cream cheese, agave nectar, the insides of the vanilla bean and the salt. Blend just until the mixture comes together as a smooth puree, but do not aerate. Meanwhile, heat the water up to a boil. As soon as the water boils remove from the heat and whisk in the Methocel. Once the Methocel is dispersed, add it to the blender and puree the contents until the mixture is homogenized, again avoid aeration.
Once the mixture is combined, pour it into a bowl over an ice bath to chill. Let the ice-cold mixture rest for at least an hour, preferably overnight before poaching the ice cream.
When ready to make the ice cream, heat a pot of water to a boil. When the water boils, shut off the heat and scoop the ice cream base. As you scoop, wipe the edges of the ice cream scoop, and then immerse the scoop and its contents into the hot water. You will see the ice cream set, and then dislodge it from the scoop. The ice cream should poach for about one minute for small scoops and longer for larger scoops. (Depending on how much ice cream you are poaching you may have to turn the heat back on to keep the water hot.)
Once the ice cream is set, remove the scoops, drain briefly on a paper towel and place into serving dishes with whatever garnishes you want. As the mixture chills the ice cream will “melt” in your dish, blending with the garnishes like and actual cold ice cream sundae.
First challenge is to get hold of methyl cellulose (also known as Methocel which is the trademark owned by Dow – BTW, they have very informative pages on food grade methyl cellulose). From Dow’s pages, it seems the SGA in the name refers to “METHOCEL Super Gelling A-Type Food Gums”. Methocell A has a DS = 1.8 and a 2% solution of this methyl cellulose sets at 50-55 °C, forming a firm gel. For a overview of Dow’s full range, check out this pdf. Click here for information about where to buy methocel (most likely in larger quantities).
For small quantities of methyl cellulose you can check out Will Goldfarb’s site (unfortunately, there’s no information about which type of methyl cellulose this is Update: It’s Dow’s F50 – a semi-firm gel forms at 62-68 °C). The Texturas series by elBulli includes Metil (with a methyl cellulose base, whatever that means), but again, I haven’t been able to find any information as to what kind of methyl cellulose this is (they do mention a gelling temperature in the range 40-60 °C however).
I’ll be happy to include further links to suppliers of methyl cellulose (and other hydrocolloids) both here and on my suppliers page if you know about any!
For those really interested, Ideas in food have several other recipes requiring methyl cellulose: hot mozarella sheets, hummus gnocchi and caramellized yoghurt gnocchi.

Sounds like a recipe that I could really do something with. Interesting idea.
Dear Sir,
I hve come across your website and congratulations for the good job.
I am the sales manager of a french company supplying an extended range of ingredients & addtivies in the field of molecular gastronomy.
We propose : alginates, calcium chloride, agar, carrageenan, gellan gum, guar gum, konjac gum, tara gum, CMC, MC, maltitol… and many other products…
If you have a look into our website, you will see that :
1- it is in french
2- it is still a test version : official version will be online in a couple of days…
not only products and kits, we provides recipes and technical informations about ptroducts properties.
I would like to know what o we have to do to be listed in your website as a supplier.
I also confirm that we can supply all over the world.
You can join me at +33 4 76 52 95 08 for any discussion.
Waiting for your feedback,
Best regards,
Yannick
[…] One challenge with the different shops is that some products come with little or no technical specification. For cellulose ethers for instance, Dow provides an extensive range to industrial customers (more on this in a previous blog post on cellulose ethers), just to give you an idea of the product range available. […]
[…] Faces – Ferrán Adrií Fuentes de Información | La Margarita Se Agita, Diario del Gourmet, Blog Khymos Otros | Texturas El […]
Hi! I was surfing and found your blog post… nice! I love your blog. 🙂 Cheers! Sandra. R.
[…] different entirely? What about modern fine dining? Do you think these chefs learned how to use methylcellulose from their grandmothers, that knowledge passed down through the generations? I could be wrong, but […]