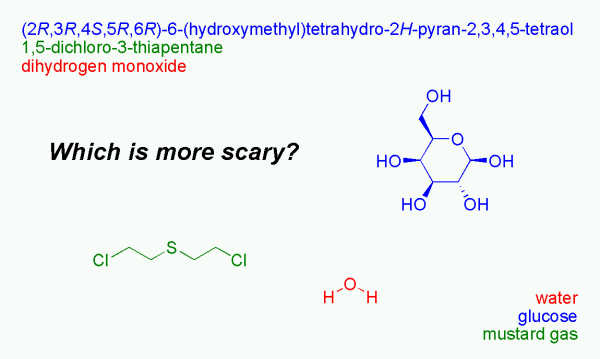
New term for molecular gastronomy?
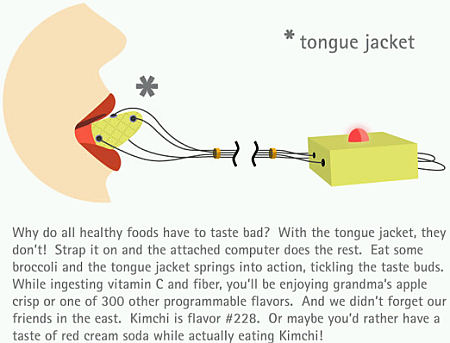
For a true multi modal experience I can imagine that restaurants and chefs who are into ORGASMIC, a new proposed acronym for science enabled cooking, will serve desserts accompanied by the orgasmatron (picture via BoingBoing). The definition and use of…